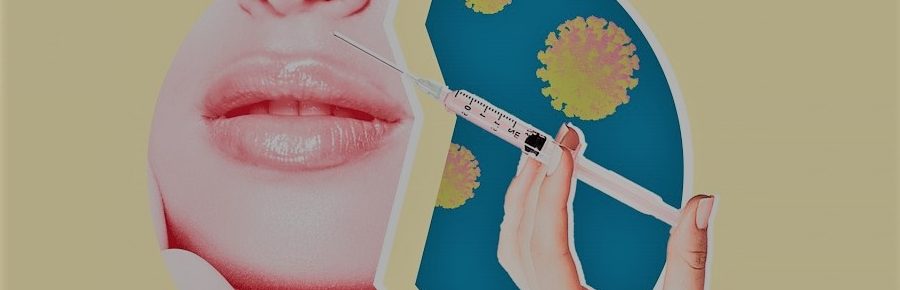
Moderna COVID Vaccine Cause Dermal Filler Reaction
FDA data reports show three participants out of 15,184 who received at least one dose of Moderna’s mRNA-1273 vaccine have developed lip or facial swelling in areas of dermal filler placement, while no participants in the placebo group experienced any filler-related adverse events. This is the first report of COVID vaccine dermal fillers reaction
Of the three affected cases none, required the use of anaphylaxis treatment or hospitalisation. As with all vaccines, swelling of the face and lips is not an uncommon side effect. Patients with a history of these complications should have epinephrine readily available. Patient safety is essential in the practice of aesthetic medicine and in their treatment, and decision making, qualified medical doctors take adverse effects seriously.
Vaccine Reaction
The Vaccines and Related Biological Products Advisory Committee (VRBPAC) reported that three people developed facial swelling. Three patients with dermal fillers have had adverse reactions to the Moderna COVID-19 vaccine. In two of these cases localised facial swelling occurred after vaccination, and in one – lip angioedema two days after vaccination. There have been no reported cases in patients receiving the Pfizer COVID-19 vaccine.
In the Moderna mRNA-1273 trial, three reactions were possibly related to dermal fillers out of 15,184 vaccine recipients. It is unknown how many subjects in the trial had previous treatment with dermal fillers.
On the foot of this reporting, the American Society for Dermatologic Surgery (ASDS) released a guidance regarding COVID-19 vaccine side effects in dermal filler treated patients. Its titled “Guidance Regarding SARS-CoV-2 mRNA Vaccine Side Effects in Dermal Filler Patients.”
The guidance provides data from the U.S. Food and Drug Administration (FDA) on reactions from the mRNA vaccine and shares factual knowledge for doctors and patients in relation to the occurrence of reactions based on the latest data available.
Dermal filler inflammatory events very uncommon occur with both hyaluronic acid and non-hyaluronic acid fillers. Existing evidence suggests these can be immune reactions triggered by viral and bacterial illness, various vaccinations and dental procedures.
Vaccine Dermal Filler Precautions
The FDA and the manufacturers of the approved COVID-19 vaccines are continuing to monitor for other similar reactions. Facilities that are providing vaccinations with either the Moderna or the Pfizer vaccines are prepared to monitor and treat patients for signs or symptoms of an allergic reaction as per recommendations from the CDC, FDA, and others.
This filler reaction is a reminder that cosmetic filler injections should be administered by qualified doctors who can ensure patient pre-treatment safety as well as manage potential complications arising from them.
These rare adverse events are transient and respond to treatments like as oral steroids and hyaluronidase.
The ASDS established a task group who plan to a multi-specialty clinical guideline on the prevention and treatment of SARS-CoV-2 mRNA vaccine-related adverse events from soft tissue fillers.
Having a history of dermal fillers should not bar someone from being vaccinated. The risks from acquiring and being infected with the COVID-19 virus far outweigh the risks from a reaction to one of the vaccines if the patient has a history of using dermal fillers.
What do you think? Did you find this blog helpful? Why not join me for meaningful and educational social media posts on Instagram I’d love to hear your comments comments@castleknockcosmetics.ie or tweet me @dr_h_kahlout. If you found this blog helpful I’d appreciate a positive review on Google Review and our Facebook page.